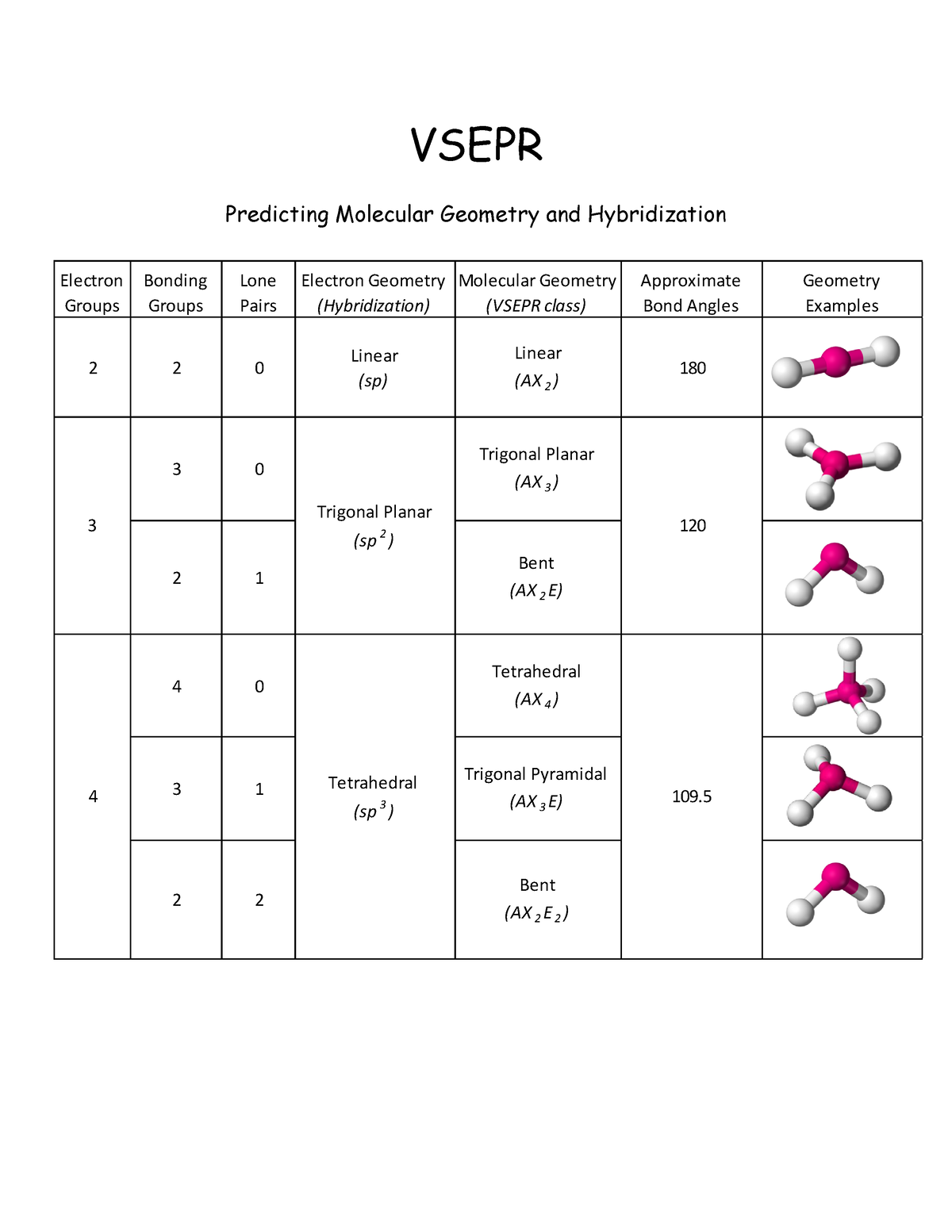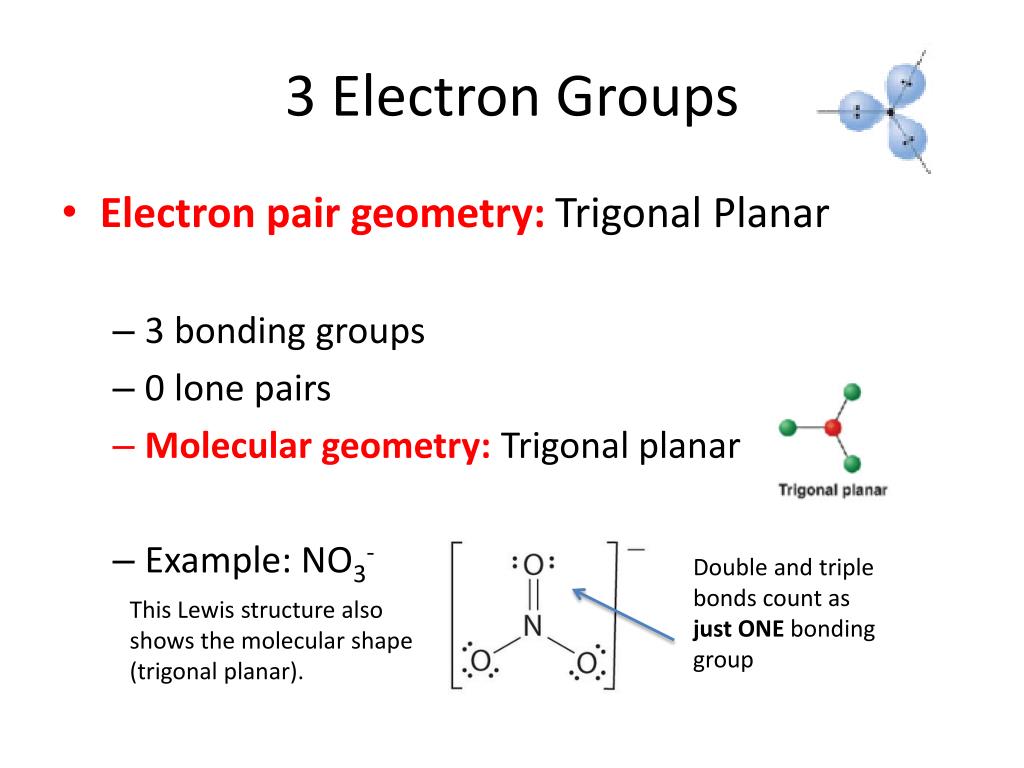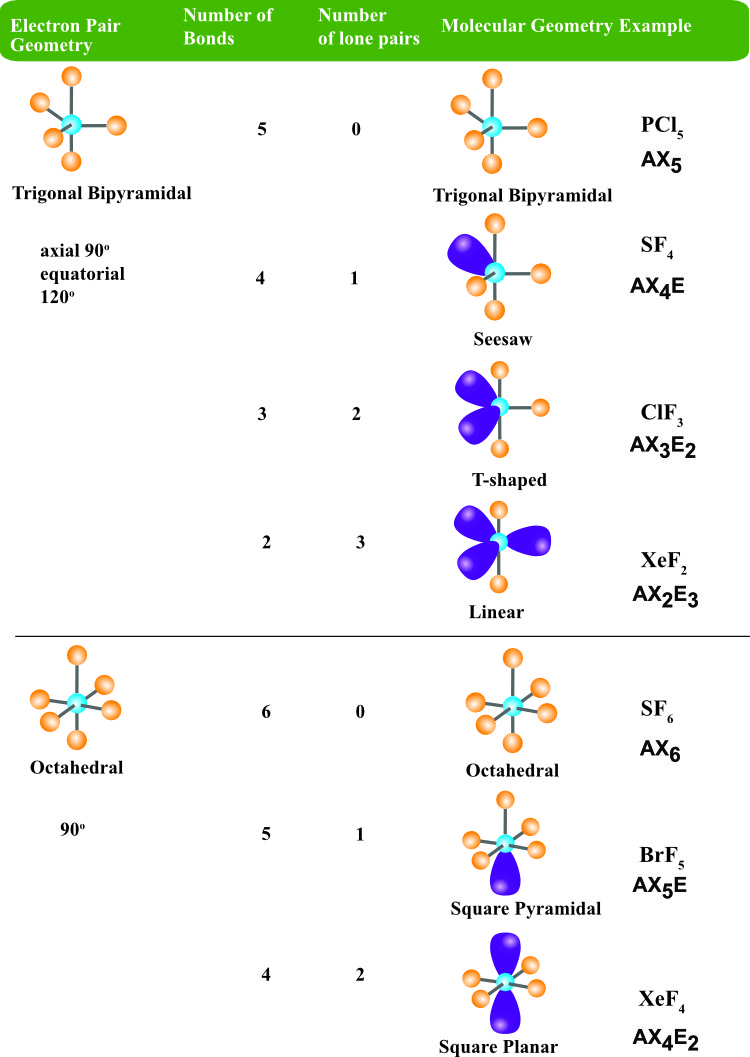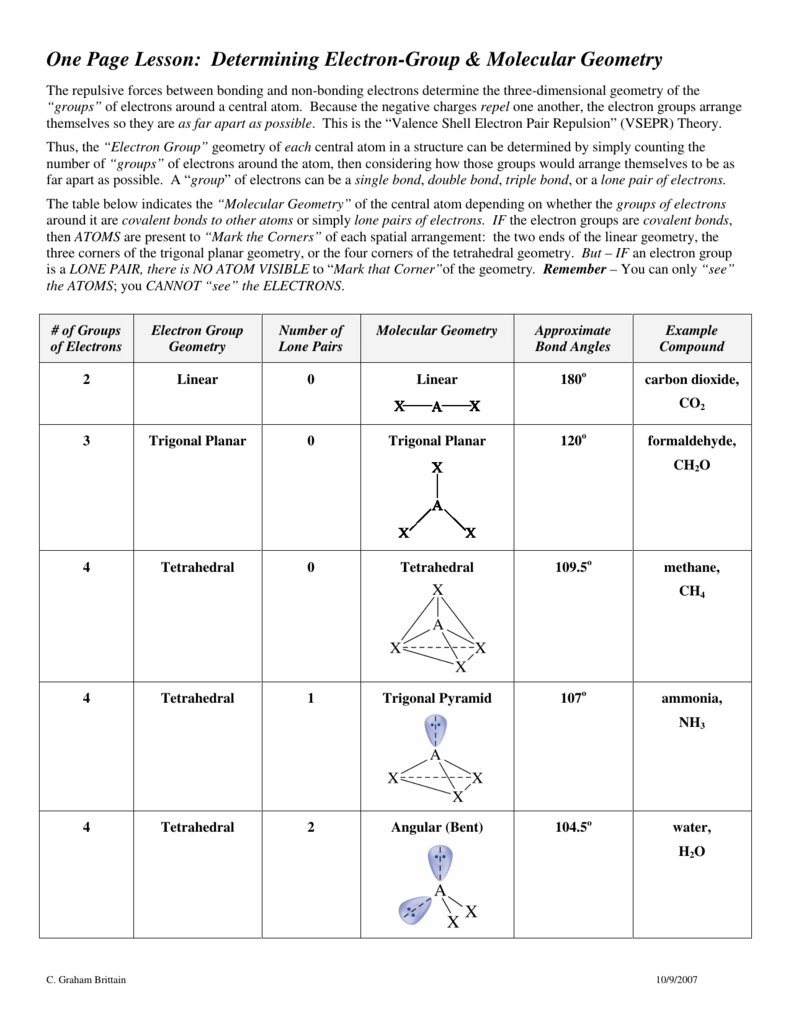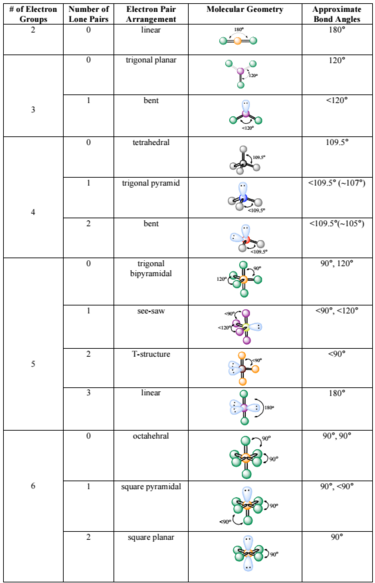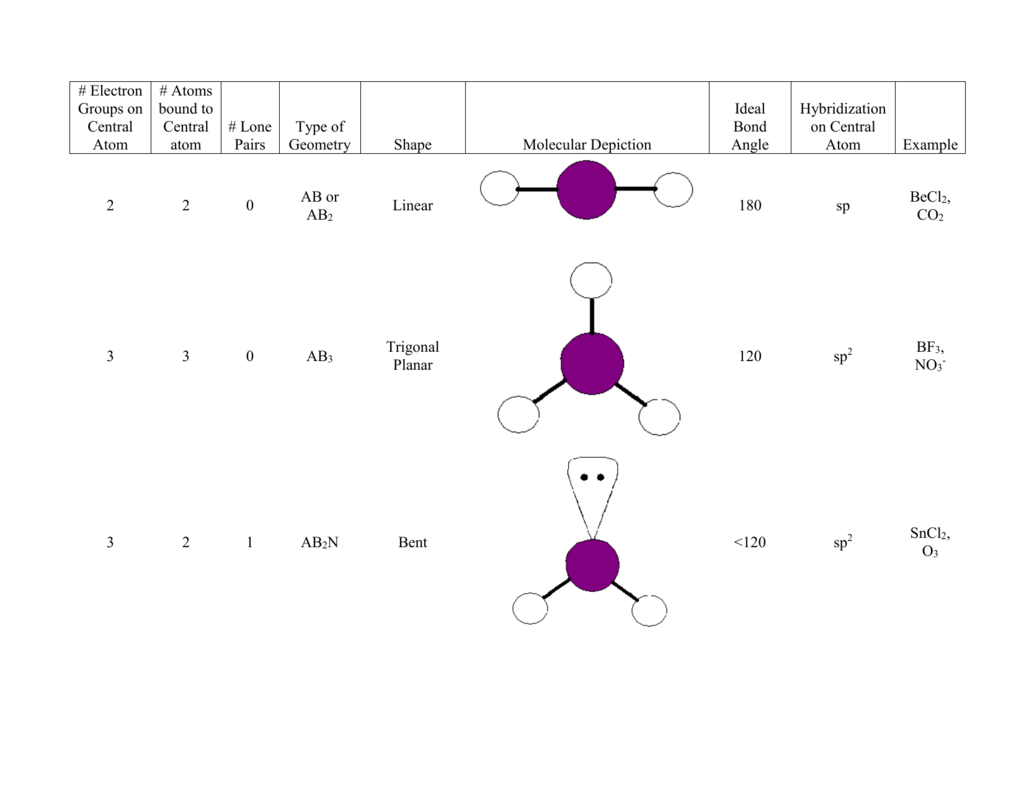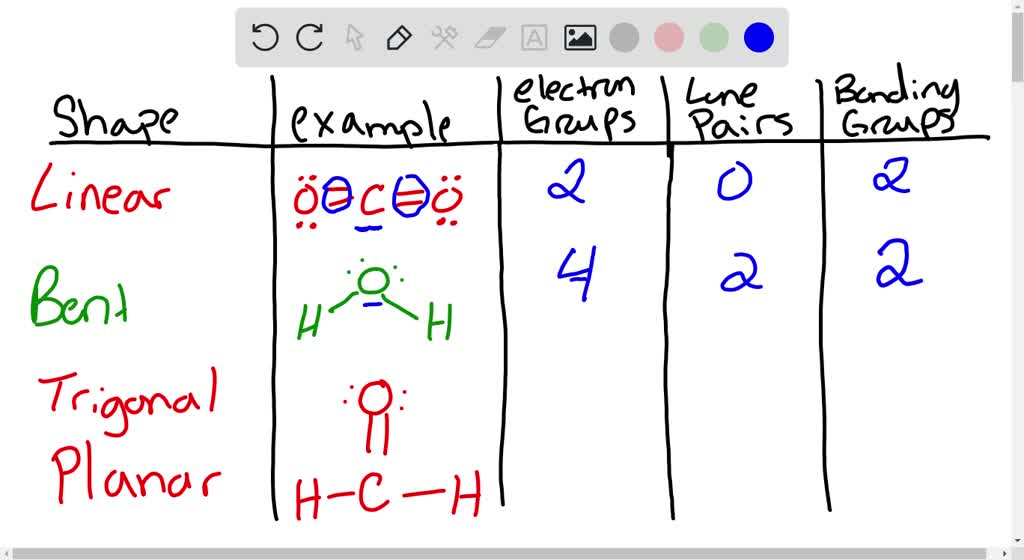
SOLVED: For each molecular geometry, list the number of total electron groups, the number of bonding groups, and the number of lone pairs on the central atom.
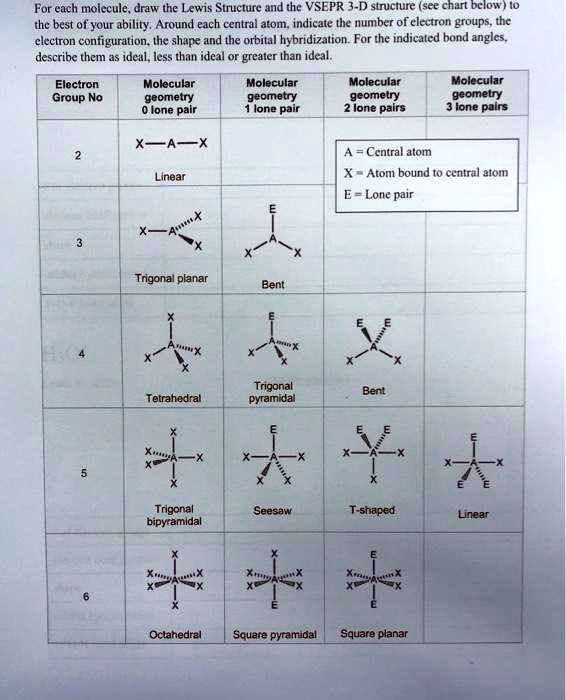
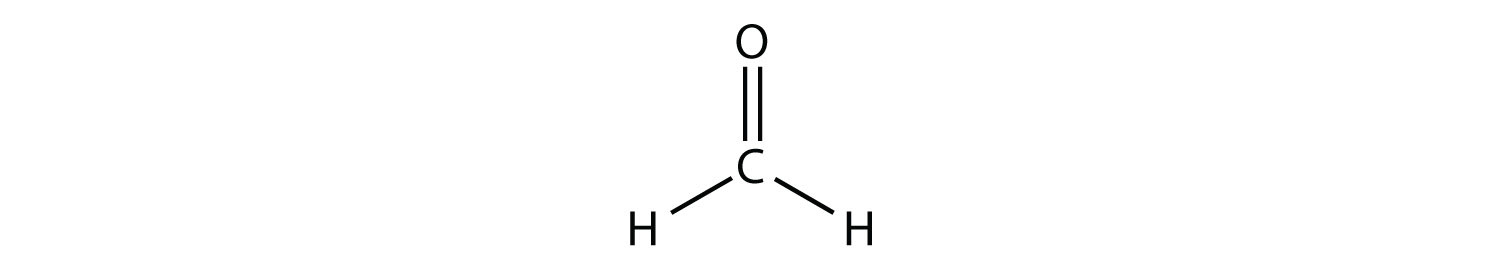
SOLVED: For each molecule; draw the Lewis Structure and the VSEPR 3-D structure (see chart below ) 0 the best of your ability. Around each central atom; indicate the number of electron

Predicting the Arrangement of Electron Groups Around the Central Atom of a Molecule | Chemistry | Study.com
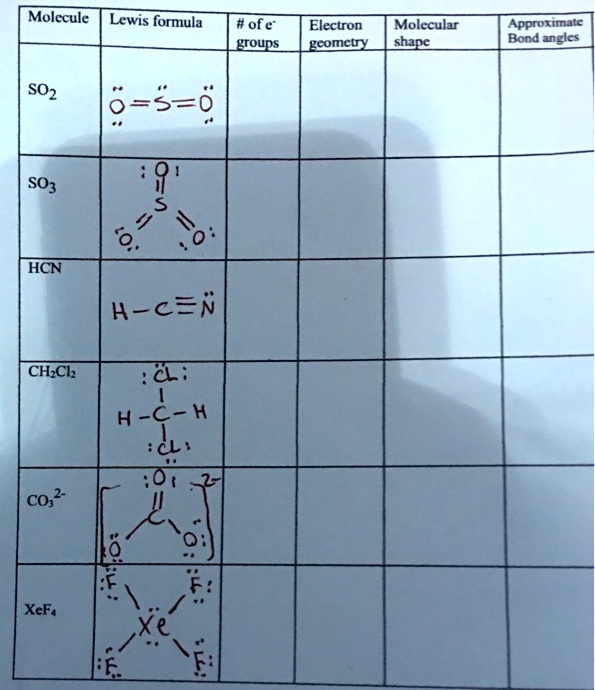
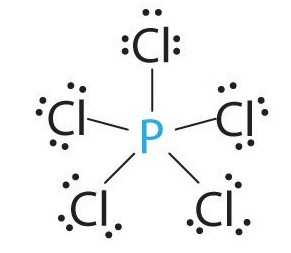
SOLVED: Molecule Lewis formula #of e" groupS Electron gcomety Molecular shape Approximate Bond angles SO2 6-5-6 SO3 8 "1 20 0 : HCN H-c=n CH-Ch 8 ; H-C-H iC: CO;" XeFs Xe F:

Valence Shell Electron Pair Repulsion (VSEPR) - Molecular & Electron Geometries of Molecules | Molecular geometry, Chemistry education, Chemistry basics